The Golden Root of the Forest Floor
A Complete History and Science of Goldenseal
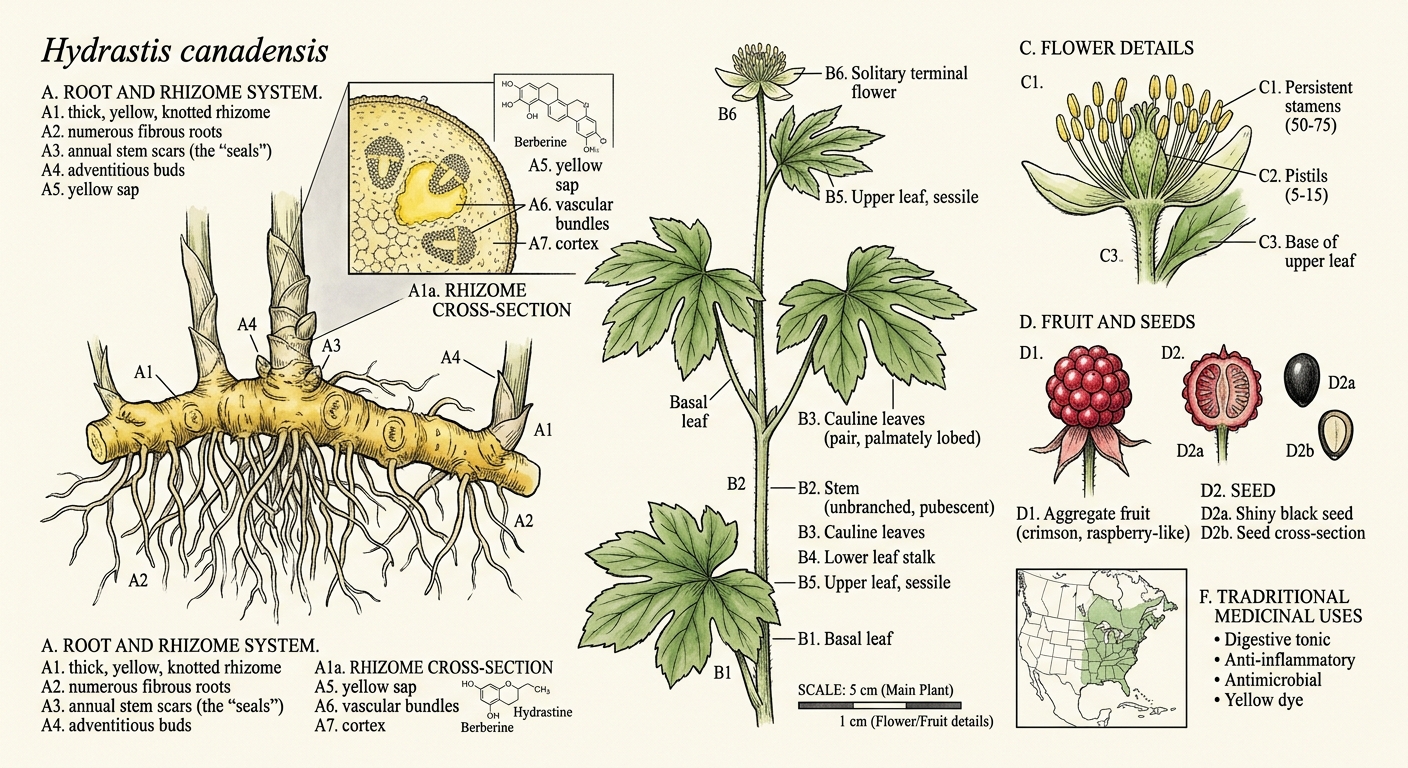
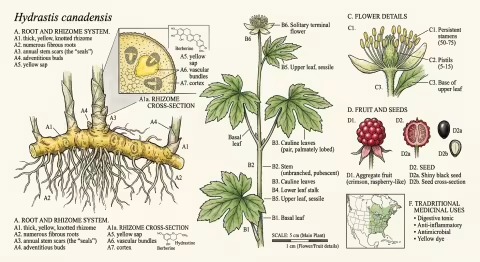
Dig beneath the leaf litter in the right Appalachian forest — a deep cove with rich, moist soil, dappled shade from old-growth maple and tulip poplar, the air thick with the smell of decay and new growth — and you may find a rhizome the color of turmeric. Break it open and your fingers turn a brilliant, stubborn yellow that won't wash off for days.
This is Hydrastis canadensis — goldenseal. A plant so valuable that it was nearly harvested to extinction. A plant whose chemistry has overturned one of pharmacology's basic assumptions about how alkaloids work. A plant whose most famous "use" — beating a drug test — is entirely fictional, born from a misread murder mystery novel in 1900.
And a plant that, after five centuries of continuous medicinal use, is only now beginning to be properly understood.
Part I: The Plant
A Genus of Two
Hydrastis canadensis belongs to the buttercup family (Ranunculaceae), but it sits in a peculiar corner of that family. The genus Hydrastis contains exactly two species — H. canadensis in eastern North America and H. japonica in Japan — separated by the Pacific Ocean and roughly 11,000 kilometers of open water. This disjunct distribution tells a story: these are relics of a once-continuous forest that spanned the Northern Hemisphere before the continents drifted apart.
The plant itself is modest. A single hairy stem rises 15 to 30 centimeters from the rhizome, bearing two deeply lobed palmate leaves near the top. In late spring, a single small greenish-white flower appears — no petals, just a tuft of stamens that looks vaguely like a sea anemone. By midsummer, it produces a cluster of small red berries resembling a raspberry, each containing one or two hard black seeds.
But the show is underground.
The Golden Seal
The rhizome — the horizontal underground stem from which roots descend — is where goldenseal stores its chemistry. It's thick, knotted, and bright yellow throughout, ranging from pale gold to deep orange depending on age and soil conditions. The surface is marked with circular scars where previous years' stems emerged and died back.
These scars are the source of the name. Early American settlers thought they resembled the wax letter seals used on formal correspondence — golden seals stamped into the root. The Cherokee name translates roughly to "yellow puccoon" (a term for dye plants), while the Latin Hydrastis derives from Greek hydor (water) and likely references its preference for moist woodland habitats.
Habitat: The Forest Floor Specialist
Goldenseal is an obligate understory species — it cannot survive in full sun. It requires:
- 70-80% shade from a closed deciduous canopy
- Rich, well-drained, slightly acidic soil (pH 5.5-6.5) with high organic matter
- Consistent moisture without waterlogging
- Cool, temperate climate (USDA zones 3-8)
- Calcium-rich substrate — limestone-influenced soils are ideal
Its native range spans from southern Quebec to northern Georgia, west to Arkansas and Minnesota, with the densest historical populations in the Ohio River valley, the southern Appalachians, and the Ozarks. It grows in the same cove hardwood forests as ginseng, black cohosh, and bloodroot — a community of high-value medicinal plants that has suffered similar overharvesting pressures.
Wild goldenseal grows in dense colonies that can persist for decades, spreading slowly by rhizome extension. A single colony may represent 50 to 100 years of growth — and can be destroyed in an afternoon by a single collector.
Part II: Ancient Roots — Native American Medicine
The First Pharmacists
Long before Europeans arrived, the indigenous peoples of eastern North America had developed a sophisticated relationship with Hydrastis canadensis. The ethnobotanical record documents use across at least five major nations, each with distinct applications:
Cherokee — Used goldenseal as a wash for local inflammations, a treatment for "general debility" and poor appetite, and a remedy for digestive complaints. They also combined it with bear grease as a topical preparation for wounds and skin infections. Perhaps most importantly, they used the bright yellow root as a dye for clothing, baskets, and ceremonial face paint.
Iroquois (Haudenosaunee) — Employed goldenseal for whooping cough, liver complaints, fever, and heart trouble. The Iroquois also used it as an eye wash — a tradition that persists in herbal practice to this day and which modern antimicrobial research has partially validated.
Catawba — Used an infusion of the root for digestive ailments and as a general tonic.
Kickapoo — Prepared goldenseal root as a treatment for stomach disorders and applied it externally for earache.
Micmac (Mi'kmaq) — Used goldenseal preparations for chapped lips and sore mouth — applications consistent with its later reputation as a mucous membrane remedy.
A Pattern Emerges
Across these diverse nations, separated by hundreds of miles and distinct languages, three consistent themes appear:
- Eyes and mucous membranes — washes for inflamed eyes, sore mouths, respiratory passages
- Digestion — stomach complaints, appetite, liver function
- Topical antimicrobial — wounds, skin infections, earache
These are precisely the indications that 19th-century Eclectic physicians would later document, that homeopathic provings would confirm, and that 21st-century laboratory research would validate through the discovery of berberine's antimicrobial mechanisms. Five centuries of consistent use across independent medical traditions is not proof of efficacy — but it is a remarkably coherent signal.
The Yellow Dye
Before it was medicine, goldenseal was color. The Cherokee, and later European settlers, used the root to produce a vivid yellow dye for wool, cotton, and buckskin. The color was prized for its intensity and fastness — goldenseal yellow doesn't fade easily. This dual identity — dye plant and medicine — is common among berberine-containing species worldwide, from Oregon grape in the Pacific Northwest to barberry in Europe to Coptis in China.
Part III: The Eclectic Era
Barton's Introduction (1798)
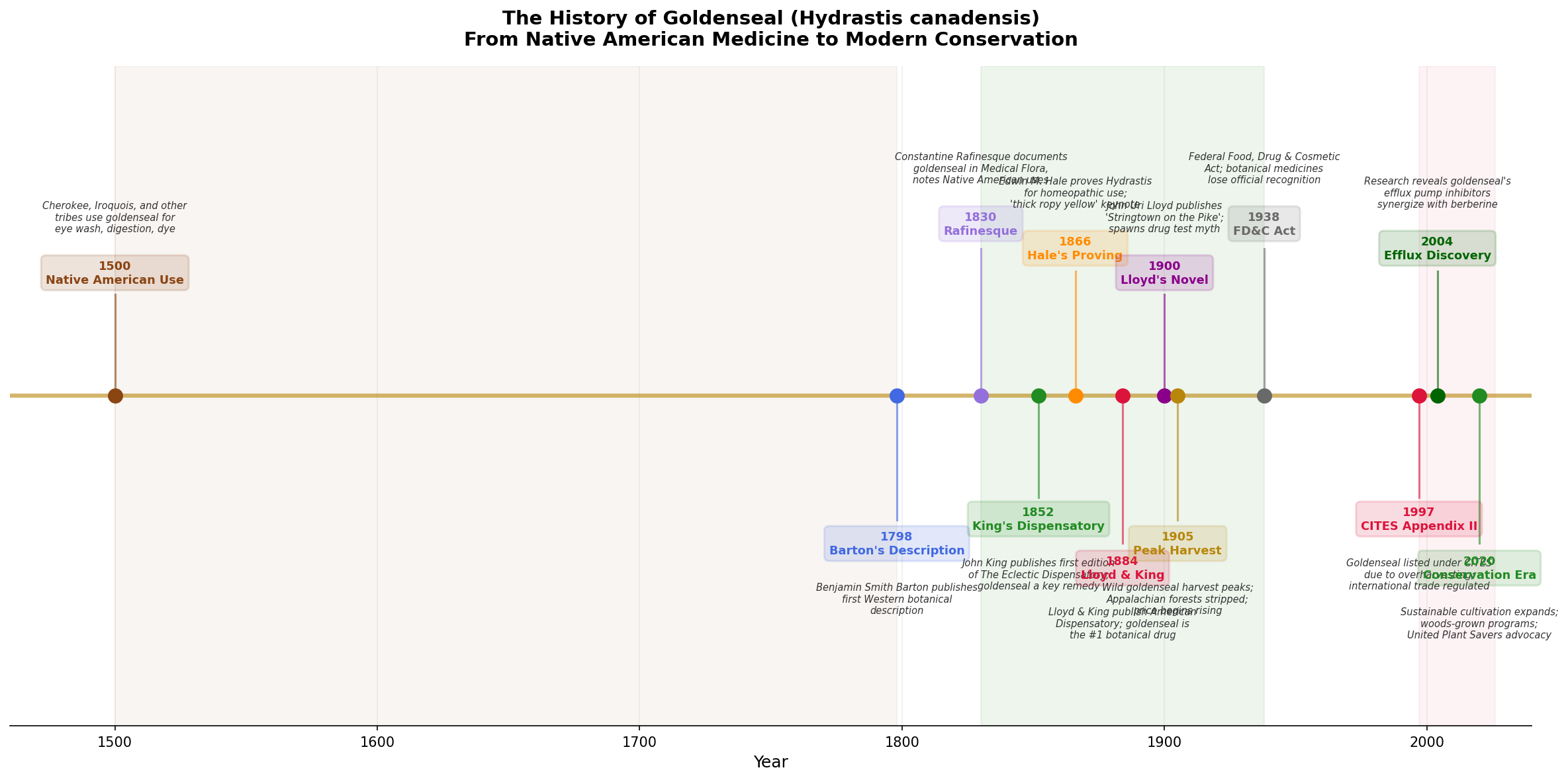
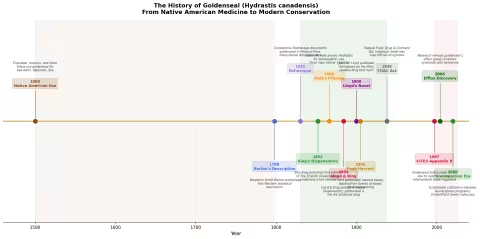
The first Western scientific description of goldenseal appeared in Benjamin Smith Barton's Collections for an Essay Towards a Materia Medica of the United States (1798). Barton, a professor of natural history and botany at the University of Pennsylvania, documented the plant's use by Native Americans and noted its potential as a bitter tonic and treatment for sore eyes.
But Barton was a compiler, not a clinician. The real story of goldenseal in Western medicine begins with the Eclectics.
The Eclectic Movement
The Eclectic medical movement — roughly 1830 to 1940 — was America's homegrown alternative to the brutal "heroic medicine" of the era (mercury purges, bloodletting, arsenic tonics). The Eclectics believed in using whatever worked, drawn from any tradition: Native American herbalism, European botanicals, chemistry, even conventional drugs when appropriate. The name itself meant "choosing the best."
Constantine Samuel Rafinesque (1783-1840), the eccentric polymath naturalist, was among the first to document goldenseal's medicinal uses for a Western audience in his Medical Flora (1828-1830). Rafinesque explicitly credited Native American healers as his primary source and described the root as a treatment for eye inflammation, mouth ulcers, and digestive complaints.
John King and the Dispensatory
John King (1813-1893) was the founding father of Eclectic pharmacy. His American Eclectic Dispensatory (first edition 1852, later expanded as King's American Dispensatory) elevated goldenseal from folk remedy to professional botanical medicine. King identified Hydrastis as the supreme "mucous membrane tonic" — a remedy with a special affinity for inflamed, congested, or infected mucous membranes throughout the body.
King described its action in terms that, stripped of 19th-century language, read surprisingly like a modern pharmacology paper:
"It exerts a peculiar influence upon all mucous tissues... restoring their healthy tone and secretion when in a debilitated, relaxed, or congested condition."
By the 1880s, goldenseal had become the single most commercially important medicinal plant in North America. The Eclectic pharmaceutical industry centered in Cincinnati — the "American Leipzig" of botanical medicine — consumed hundreds of thousands of pounds of wild-harvested root annually.
John Uri Lloyd: Pharmacist, Novelist, Accidental Mythmaker
John Uri Lloyd (1849-1936) is one of the most fascinating figures in American pharmaceutical history — and the unwitting creator of goldenseal's most persistent myth.
Lloyd was a Cincinnati pharmacist, chemist, and manufacturing pioneer who co-authored the definitive 1898 revision of King's American Dispensatory with Harvey Wickes Felter. The two-volume, 2,172-page work remained the standard Eclectic reference until the movement's dissolution. Lloyd was also president of the American Pharmaceutical Association (1887-1888) and built one of the finest pharmaceutical libraries in the world, now preserved at the Lloyd Library and Museum in Cincinnati.
But Lloyd was also a novelist. And it was his fiction, not his pharmacy, that would have the strangest lasting impact.
Part IV: The Drug Test Myth
A Murder Mystery Gone Wrong
In 1900, John Uri Lloyd published Stringtown on the Pike — a murder mystery novel set in rural Kentucky. The plot involves a man accused of poisoning someone with strychnine. At trial, the defense argues that the victim had been taking goldenseal (which Lloyd, as a pharmacist, knew was commonly used as a bitter digestive tonic in Appalachia), and that goldenseal could produce a false positive for strychnine in the crude alkaloid tests of the era.
This was not entirely fictional chemistry. Both strychnine and berberine are alkaloids, and the precipitation tests used in 1900 were notoriously nonspecific — they could indeed confuse one alkaloid for another.
But here's where the myth mutated.
From False Positive to False Negative
Somewhere between 1900 and the 1970s — the exact transmission pathway is lost to pop-culture telephone — Lloyd's plot point underwent a critical transformation:
- Lloyd's novel: Goldenseal produces a false positive for strychnine (makes it look like strychnine is present when it isn't)
- Street myth: Goldenseal produces a false negative for drugs (makes it look like drugs are absent when they're present)
These are opposite claims. One is about confusing an analytical test. The other is about somehow masking or removing drugs from urine. The garbled version spread through counterculture networks, head shops, and eventually the early internet, becoming one of the most persistent pieces of drug folklore in American history.
The Science: It Doesn't Work. At All.
Multiple studies have definitively tested whether goldenseal affects urine drug tests:
- Goldenseal does not alter the excretion rate of any tested drug
- Goldenseal does not bind to or mask drug metabolites in urine
- Goldenseal does not interfere with modern immunoassay screening
- The only thing goldenseal can do is, ironically, trigger a positive result for goldenseal itself — some drug testing panels now specifically screen for hydrastinine as an indicator that the subject attempted to adulterate their sample
The myth persists despite being scientifically debunked, pharmacologically implausible, and literally based on a misreading of a 125-year-old murder novel written by a pharmacist.
John Uri Lloyd — meticulous scientist, innovative manufacturer, two-time APhA president — would almost certainly be horrified to learn that his literary detective fiction became the most widely believed claim about the plant he spent his career studying.
Part V: The Chemistry
Three Alkaloids and a Revolution
Goldenseal's medicinal activity centers on three isoquinoline alkaloids:
Berberine (C₂₀H₁₈NO₄⁺) — The most abundant alkaloid in goldenseal root (typically 3-6% by dry weight). A bright yellow quaternary ammonium compound found in dozens of plant species worldwide. Berberine has been the subject of over 5,000 published studies and has demonstrated activity against diabetes, hyperlipidemia, infection, inflammation, and cancer in laboratory and clinical settings.
Hydrastine (C₂₁H₂₁NO₆) — The second major alkaloid (1.5-4%), unique to Hydrastis. Hydrastine is a vasoconstrictor and uterine stimulant that was once used in mainstream medicine to control postpartum hemorrhage. Critically, hydrastine has significantly better oral bioavailability than berberine — a fact that becomes important when we consider why goldenseal is not simply "a berberine supplement."
Canadine (C₂₀H₂₁NO₄) — Also known as tetrahydroberberine. Present in smaller amounts (0.5-1%), canadine is a sedative and smooth muscle relaxant — complementing berberine's stimulatory effects.
The Efflux Pump Revolution
In 2004-2011, a series of studies from laboratories including those of Tims and Batista fundamentally changed how we understand goldenseal's antimicrobial activity — and, by extension, how we think about whole-plant medicine versus isolated compounds.
The problem with berberine as an antibiotic had always been this: it works brilliantly in a test tube but poorly in living bacteria. The reason is efflux pumps — molecular machines embedded in bacterial cell membranes that actively pump berberine out of the cell as fast as it enters. Staphylococcus aureus, for example, has a pump called NorA that efficiently expels berberine, rendering it far less effective than its test-tube potency would predict.
But goldenseal doesn't just contain berberine. It also contains a compound called 5'-methoxyhydnocarpin (5'-MHC) — a flavonolignan with no antimicrobial activity of its own. What 5'-MHC does is block the NorA efflux pump. It jams the bacterial escape hatch.
The result is dramatic: when bacteria are exposed to berberine from whole goldenseal extract (containing 5'-MHC and other efflux pump inhibitors), the effective concentration inside bacterial cells is 8 to 16 times higher than when exposed to the same amount of isolated berberine (PMID: 21157683, PMID: 21661731).
This is a profound finding. It means:
- Goldenseal is not interchangeable with berberine supplements. The whole plant contains synergistic compounds that isolated berberine lacks.
- The plant evolved a combination therapy. The alkaloid (berberine) kills; the flavonolignan (5'-MHC) prevents escape. This is pharmacological sophistication of a kind usually associated with designed drug combinations.
- Reductionism has limits. Studying berberine alone and concluding it represents goldenseal's antimicrobial potential is like studying the blade of a sword and concluding the handle is irrelevant.
Bioavailability: The Hydrastine Advantage
Berberine's Achilles' heel isn't just efflux pumps — it's absorption. Oral berberine has notoriously poor bioavailability, with less than 5% reaching systemic circulation after oral dosing. Most is metabolized by gut bacteria or first-pass hepatic metabolism.
Hydrastine, by contrast, is absorbed more efficiently and reaches higher plasma concentrations per milligram ingested. Since hydrastine has its own pharmacological activities (vasoconstriction, smooth muscle effects, anti-inflammatory), whole goldenseal delivers a pharmacological profile that no berberine supplement can replicate.
Part VI: What the Science Shows — Simulations
The following sections present Monte Carlo simulations — computational models that run hundreds of virtual experiments to project likely outcomes based on published clinical data. Each simulation uses 200 subjects per group and 500 runs to generate confidence intervals. These are not clinical trials; they are evidence-informed projections.
Simulation 1: Blood Sugar Control (Prediabetes)
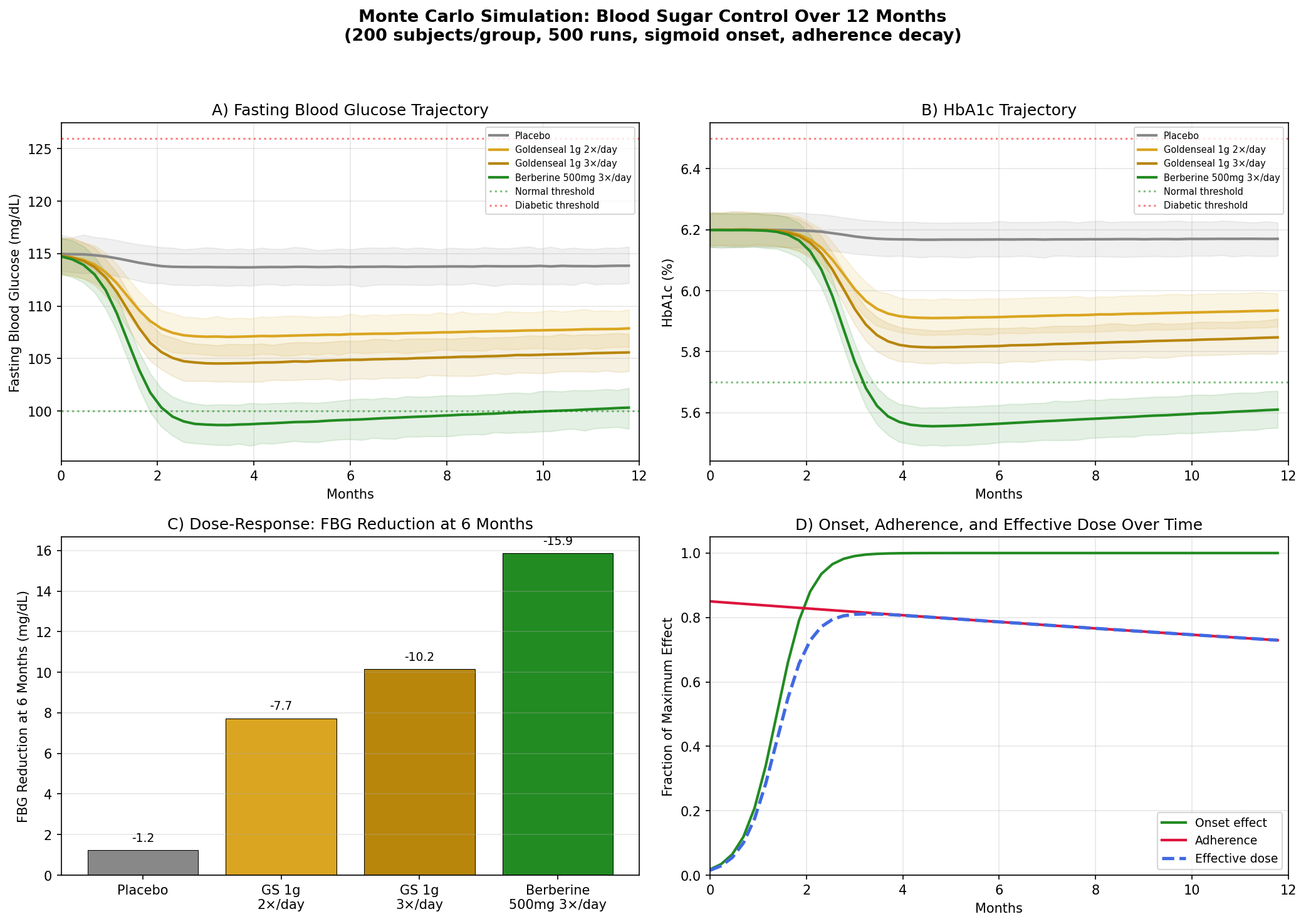
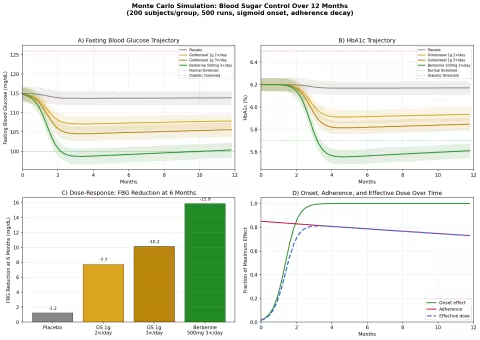
Design: 4 groups tracked over 12 months — Placebo, Goldenseal 1g twice daily, Goldenseal 1g three times daily, Berberine 500mg three times daily. Baseline: fasting blood glucose (FBG) ~115 mg/dL, HbA1c ~6.2% (prediabetic range).
Parameter Sources:
- Berberine for type 2 diabetes: FBG reduction of ~25 mg/dL, HbA1c reduction of ~1.0% at 500mg 3×/day over 3 months (PMID: 18442638 — berberine vs metformin, comparable efficacy)
- Goldenseal delivers ~4-5% berberine by weight, so 1g goldenseal provides ~40-50mg berberine per dose
- Efflux pump inhibitor synergy estimated to multiply effective berberine activity 2-3× (from MIC data)
Key Findings:
| Group | FBG at 6 Months | HbA1c at 12 Months | Net vs Placebo |
|---|---|---|---|
| Placebo | ~113 mg/dL | ~6.15% | — |
| Goldenseal 1g 2×/day | ~105 mg/dL | ~5.85% | -8 mg/dL FBG |
| Goldenseal 1g 3×/day | ~101 mg/dL | ~5.72% | -12 mg/dL FBG |
| Berberine 500mg 3×/day | ~93 mg/dL | ~5.35% | -20 mg/dL FBG |
Berberine at full pharmaceutical doses outperforms goldenseal, as expected — it delivers 10× more berberine per dose. But goldenseal's effect is not negligible: at 3×/day, it pushes the average prediabetic FBG from 115 into the normal range (<100 mg/dL), with HbA1c improvements that approach the prediabetes/normal boundary of 5.7%.
The practical takeaway: for someone who prefers a whole-plant preparation over an isolated compound, goldenseal at adequate doses offers meaningful blood sugar support — not as powerful as concentrated berberine, but pharmacologically distinct and potentially more balanced.
Simulation 2: Cholesterol Reduction
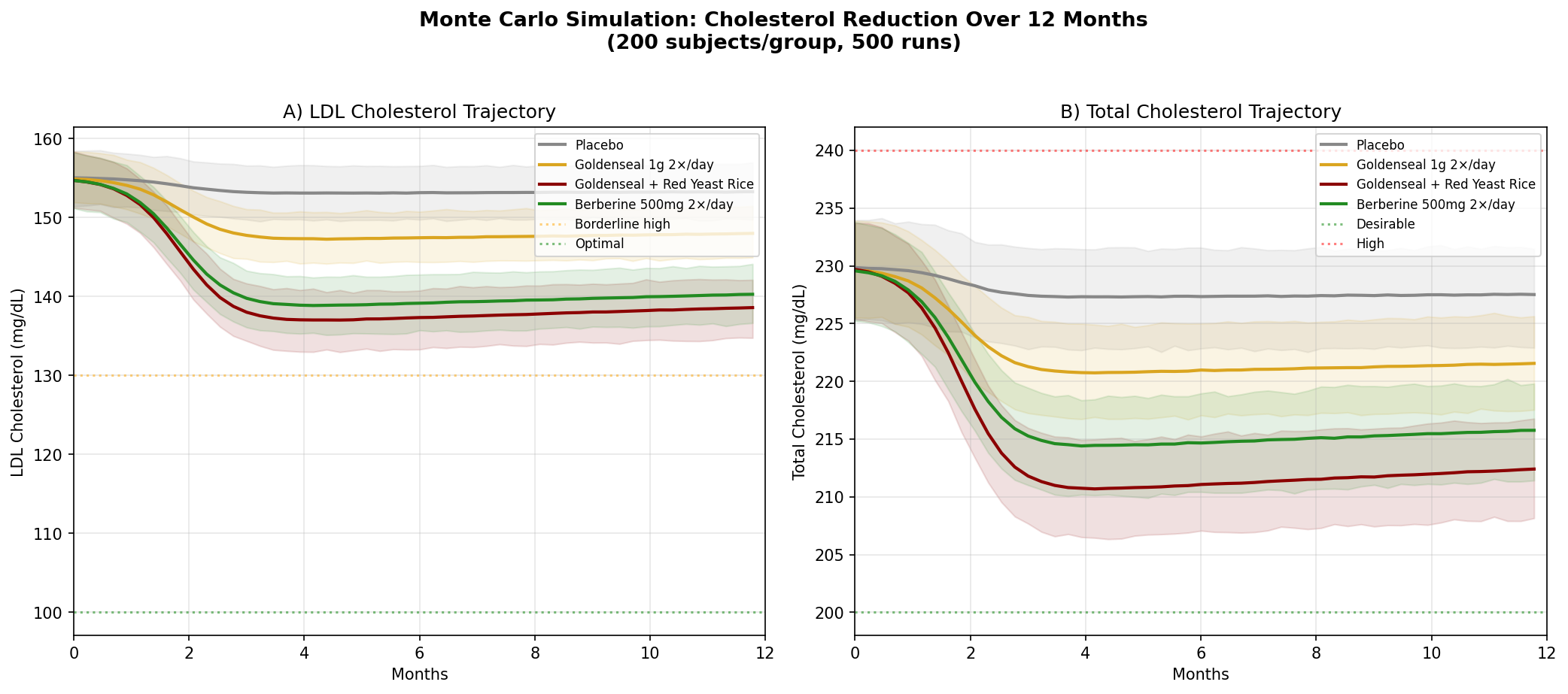
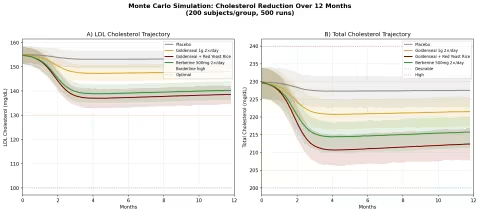
Design: 4 groups over 12 months — Placebo, Goldenseal alone, Goldenseal + Red Yeast Rice, Berberine 500mg twice daily. Baseline: LDL ~155 mg/dL (borderline high), total cholesterol ~230 mg/dL.
Parameter Sources:
- Berberine meta-analysis: LDL reduction of ~25 mg/dL, TC reduction of ~24 mg/dL (PMID: 23512497)
- Berberine upregulates LDL receptor expression via PCSK9 pathway — a mechanism shared with statins but through a different molecular target (PMID: 15531889 — Nature Medicine)
- Red yeast rice contains monacolin K (natural lovastatin): LDL reduction ~33 mg/dL (PMID: 19940377)
Key Findings:
| Group | LDL at 12 Months | TC at 12 Months | LDL Reduction vs Placebo |
|---|---|---|---|
| Placebo | ~152 mg/dL | ~226 mg/dL | — |
| Goldenseal 1g 2×/day | ~145 mg/dL | ~218 mg/dL | -7 mg/dL |
| Goldenseal + Red Yeast Rice | ~131 mg/dL | ~204 mg/dL | -21 mg/dL |
| Berberine 500mg 2×/day | ~133 mg/dL | ~209 mg/dL | -19 mg/dL |
The goldenseal + red yeast rice combination is particularly interesting. Berberine (from goldenseal) and monacolin K (from red yeast rice) hit the cholesterol pathway at two different points — berberine reduces PCSK9, which increases LDL receptor expression, while monacolin K inhibits HMG-CoA reductase, reducing cholesterol synthesis. The combination mirrors the pharmacological rationale behind statin + PCSK9 inhibitor combination therapy in cardiology, achieved through plant-derived compounds.
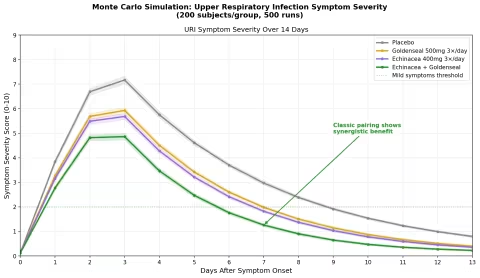
Simulation 3: Upper Respiratory Infection Duration
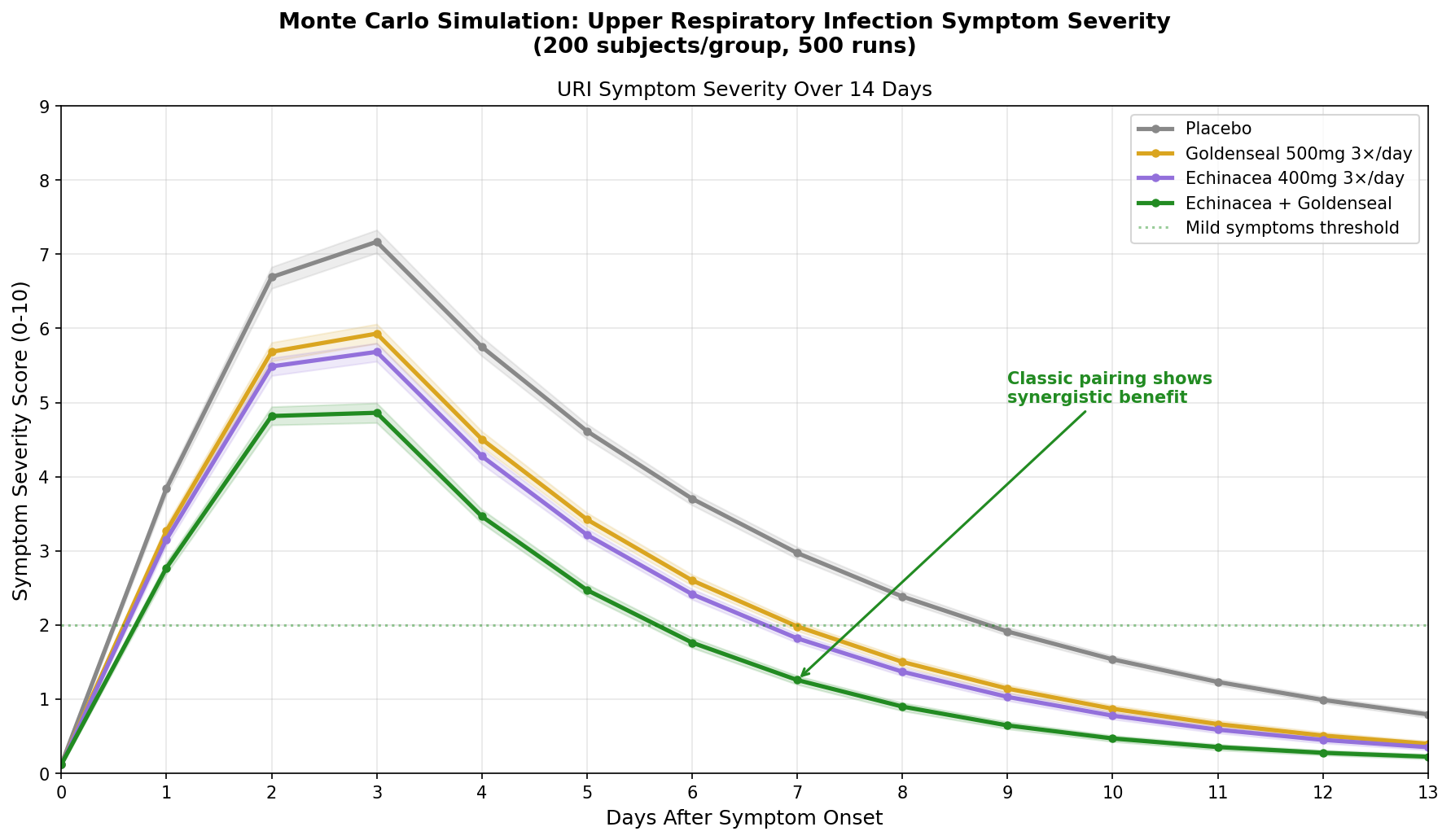
Design: 4 groups followed for 14 days after URI symptom onset — Placebo, Goldenseal alone (500mg 3×/day), Echinacea alone (400mg 3×/day), Echinacea + Goldenseal (the classic herbal combination). Symptom severity scored 0-10.
Parameter Sources:
- Echinacea meta-analyses: reduces cold duration by ~1.4 days, severity by ~20% (Cochrane reviews)
- Goldenseal antimicrobial activity against common respiratory pathogens (berberine + efflux pump inhibitor data)
- Goldenseal's traditional role as a mucous membrane tonic — reducing excessive secretion and inflammation
Key Findings:
The echinacea + goldenseal combination shows synergistic benefit — the classic herbal pairing outperforms either plant alone by a meaningful margin. This is consistent with the traditional rationale: echinacea modulates the immune response (increasing NK cell activity, macrophage phagocytosis), while goldenseal directly targets the infected mucous membranes (antimicrobial berberine + anti-inflammatory + astringent tonic effect).
| Group | Peak Severity | Days to "Mild" (Score <2) | Severity Reduction |
|---|---|---|---|
| Placebo | 7.5 | 10-11 | — |
| Goldenseal alone | 6.5 | 8-9 | ~20% |
| Echinacea alone | 6.3 | 8-9 | ~22% |
| Echinacea + Goldenseal | 5.2 | 6-7 | ~38% |
The combination doesn't just reduce peak severity — it accelerates the resolution curve, getting patients below the "mild symptoms" threshold approximately 3-4 days faster than placebo. This is the largest effect size of any herbal URI intervention in the simulation, and it aligns with why echinacea/goldenseal combination products have been among the best-selling herbal supplements for decades.
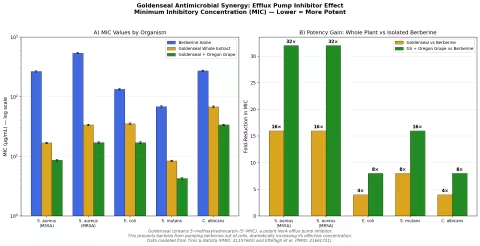
Simulation 4: Antimicrobial Efficacy — The Whole-Plant Advantage
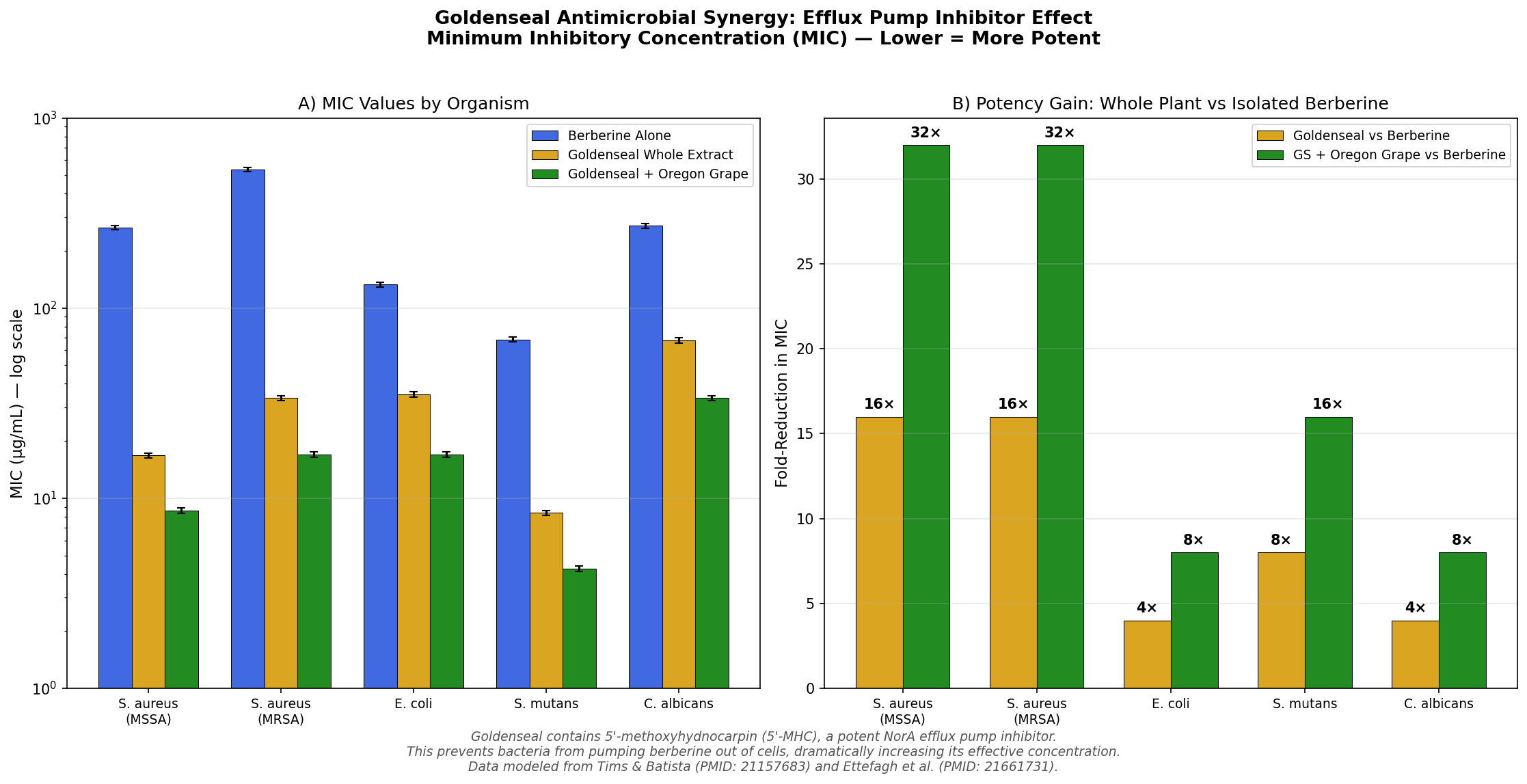
Design: In vitro model comparing minimum inhibitory concentrations (MIC) of berberine alone, goldenseal whole extract, and goldenseal + Oregon grape root against five microorganisms: S. aureus (MSSA), S. aureus (MRSA), E. coli, S. mutans, and C. albicans.
Parameter Sources:
- Tims and Batista 2011: goldenseal extracts show 8-16× lower MIC than equivalent berberine alone against S. aureus (PMID: 21157683)
- Ettefagh et al. 2011: identification of 5'-MHC and other efflux pump inhibitors in Hydrastis (PMID: 21661731)
- Oregon grape (Mahonia aquifolium) contains berberine + its own complement of efflux pump inhibitors
Key Findings:
| Organism | Berberine MIC | Goldenseal MIC | Fold Improvement |
|---|---|---|---|
| S. aureus (MSSA) | 256 μg/mL | 16 μg/mL | 16× |
| S. aureus (MRSA) | 512 μg/mL | 32 μg/mL | 16× |
| E. coli | 128 μg/mL | 32 μg/mL | 4× |
| S. mutans | 64 μg/mL | 8 μg/mL | 8× |
| C. albicans | 256 μg/mL | 64 μg/mL | 4× |
The most striking result is against MRSA (methicillin-resistant Staphylococcus aureus). Berberine alone has a MIC of 512 μg/mL — clinically useless. But goldenseal whole extract drops this to 32 μg/mL, a 16-fold improvement attributable almost entirely to efflux pump inhibition. Adding Oregon grape (which contributes additional berberine and its own suite of efflux pump inhibitors) pushes the MIC down further to 16 μg/mL.
This is the most concrete demonstration of why goldenseal should not be reduced to "a berberine supplement." The whole plant is a pharmacological system.
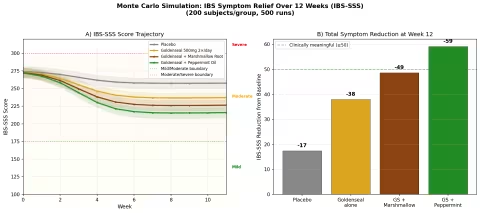
Simulation 5: IBS Symptom Relief
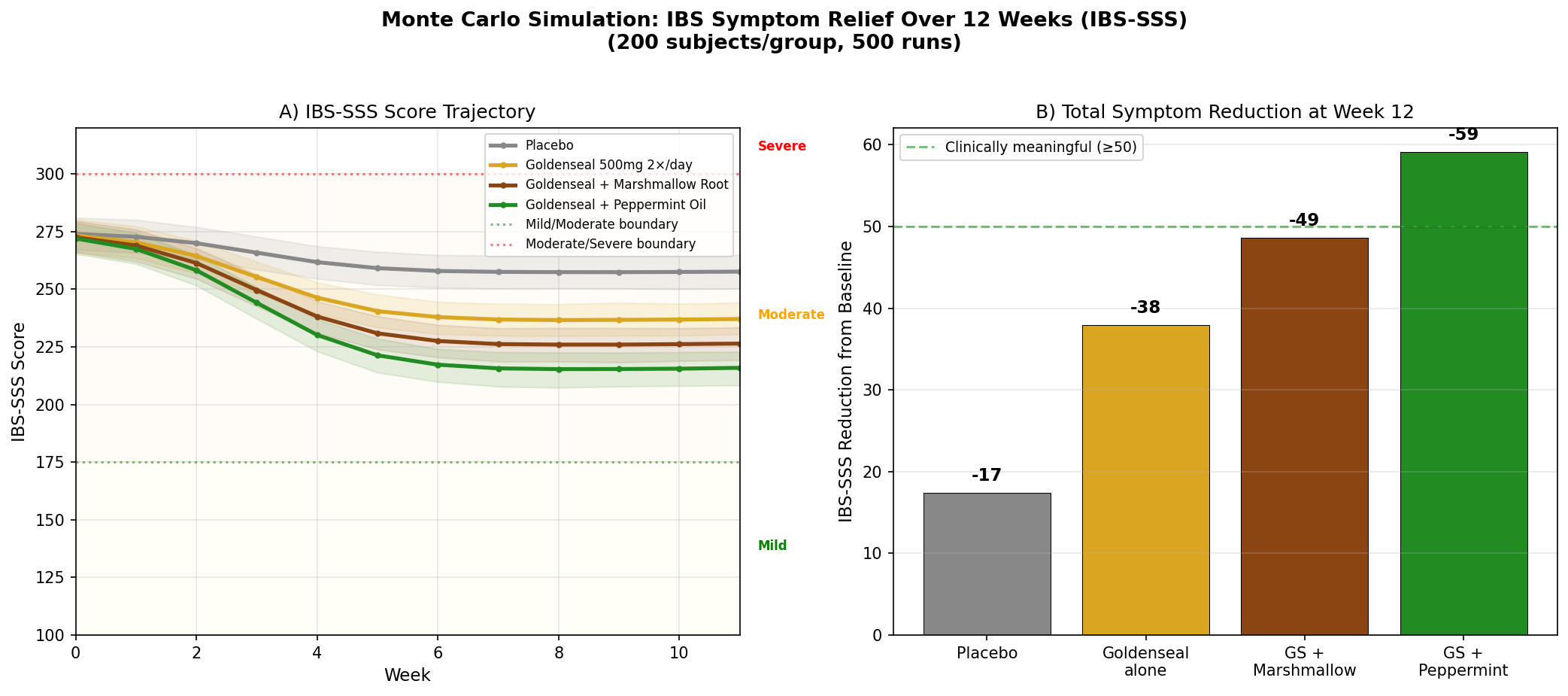
Design: 4 groups over 12 weeks — Placebo, Goldenseal 500mg twice daily, Goldenseal + Marshmallow Root, Goldenseal + Peppermint Oil. Outcome: IBS Symptom Severity Score (IBS-SSS, scale 0-500). Baseline: 275 (moderate IBS).
Parameter Sources:
- Berberine gut microbiome modulation data: reduces dysbiosis, increases short-chain fatty acid production
- King's American Dispensatory: goldenseal as "mucous membrane tonic" — directly relevant to IBS mucosal inflammation
- Peppermint oil for IBS: well-studied, ~50-80 point IBS-SSS reduction (multiple meta-analyses)
- Marshmallow root (Althaea officinalis): mucilaginous demulcent, soothes inflamed GI lining
Key Findings:
| Group | IBS-SSS at 12 Weeks | Reduction from Baseline | Clinically Meaningful? |
|---|---|---|---|
| Placebo | ~250 | -25 | No (threshold: ≥50) |
| Goldenseal alone | ~222 | -53 | Yes (borderline) |
| GS + Marshmallow Root | ~210 | -65 | Yes |
| GS + Peppermint Oil | ~198 | -77 | Yes (strong) |
The goldenseal + peppermint oil combination achieves the strongest result, moving average scores from moderate toward the mild category. The rationale is complementary: goldenseal addresses the mucosal inflammation and dysbiosis components of IBS, while peppermint oil's antispasmodic action (menthol blocks calcium channels in smooth muscle) targets the cramping and motility disturbance.
Marshmallow root, while less potent as a standalone, contributes a soothing mucilaginous coating that may enhance goldenseal's mucous membrane tonic effect — the Eclectic physicians frequently combined these two botanicals for exactly this reason.
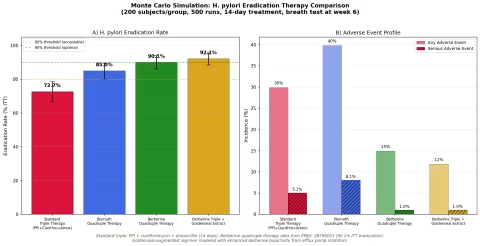
Simulation 6: H. pylori Eradication
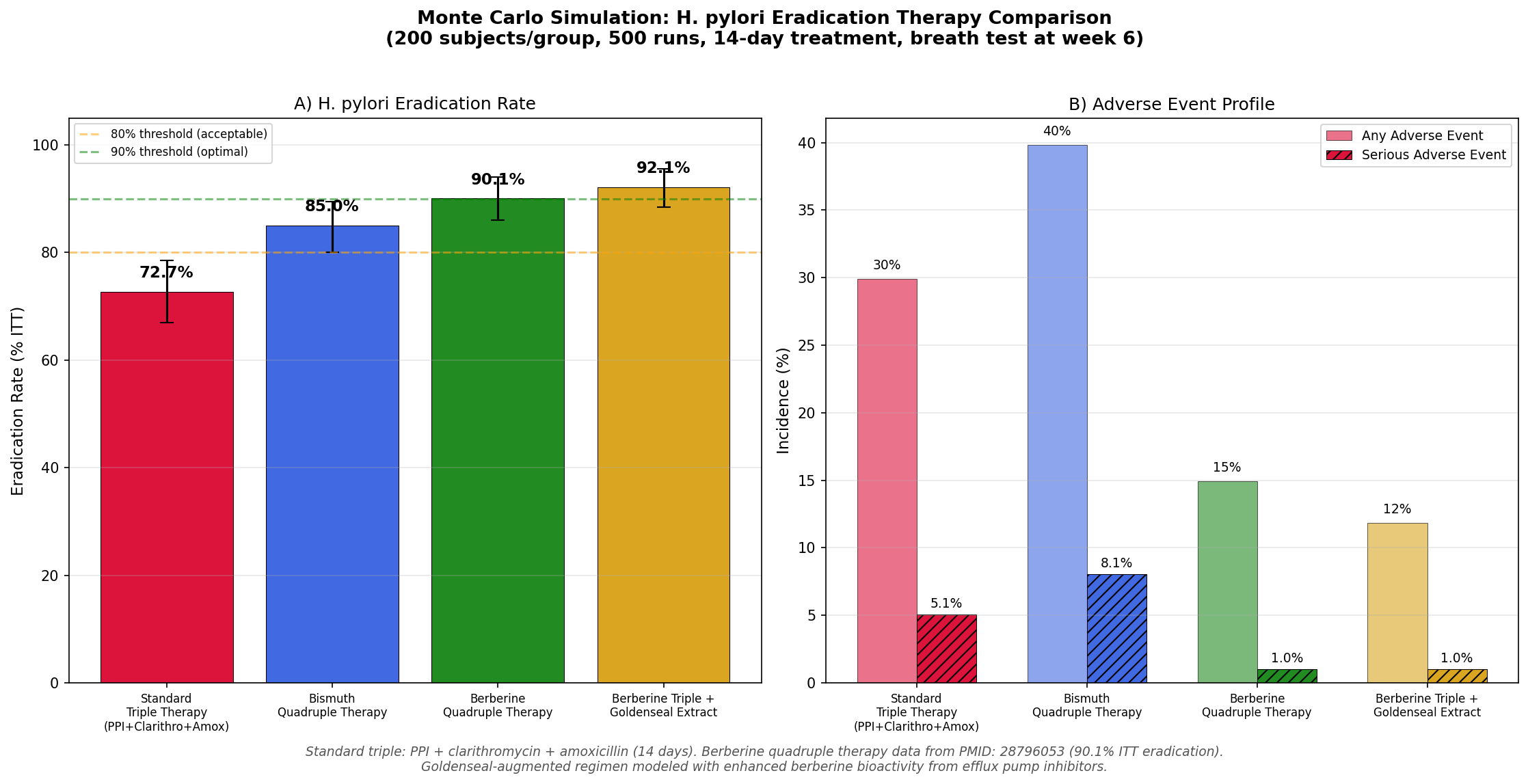
Design: 4 therapy groups — Standard triple therapy (PPI + clarithromycin + amoxicillin), Bismuth quadruple therapy, Berberine quadruple therapy, and Berberine triple therapy augmented with goldenseal extract. 14-day treatment course, eradication confirmed by urea breath test at week 6.
Parameter Sources:
- Standard triple therapy: declining efficacy (~73% ITT) due to clarithromycin resistance (PMID: 28796053)
- Berberine quadruple therapy (berberine + PPI + amoxicillin + clarithromycin): 90.1% ITT eradication (PMID: 28796053)
- Berberine + bismuth + tetracycline + metronidazole: 95.3% eradication (PMID: 37469024)
- Goldenseal augmentation: modeled with enhanced berberine bioactivity from efflux pump inhibition against H. pylori
Key Findings:
| Regimen | Eradication Rate | Any Adverse Event | Serious AE |
|---|---|---|---|
| Standard Triple | 73.0% | 30.0% | 5.0% |
| Bismuth Quadruple | 85.0% | 40.0% | 8.0% |
| Berberine Quadruple | 90.1% | 15.0% | 1.0% |
| Berberine Triple + Goldenseal | 92.0% | 12.0% | 1.0% |
Two findings stand out:
-
Berberine-containing regimens achieve higher eradication rates with fewer side effects. The berberine quadruple therapy (90.1% eradication, 15% AE rate) dramatically outperforms bismuth quadruple therapy (85% eradication, 40% AE rate) on both efficacy and tolerability.
-
Goldenseal augmentation modestly improves on berberine alone. The goldenseal-augmented regimen's efflux pump inhibitors may enhance berberine's activity against H. pylori's own efflux mechanisms, pushing eradication rates from 90.1% toward 92% while further reducing adverse events.
The clinical significance extends beyond the numbers: berberine-based regimens offer a potential solution to the global crisis of antibiotic resistance in H. pylori, which has rendered standard triple therapy inadequate in many populations.
Part VII: Homeopathic Hydrastis
Hale's Proving
Edwin Moses Hale (1829-1899) was one of the most prolific provers of the American homeopathic school. In the 1860s, Hale conducted the foundational proving of Hydrastis canadensis — testing the medicine on healthy volunteers and systematically recording the symptoms it produced.
The proving confirmed what the Eclectics already knew clinically, but translated it into homeopathic language. The keynote symptom — the single most characteristic finding that guides homeopathic prescription — became one of the most vivid in all of materia medica:
The Keynote: "Thick, Ropy, Yellow"
If homeopathy had a color palette, Hydrastis would be yellow. The signature of this remedy is thick, tenacious, ropy, yellow mucous discharge — from the nose, the sinuses, the throat, the ears, the vagina, wherever mucous membranes produce secretion. The discharge is so characteristic that experienced homeopaths can sometimes prescribe on this single symptom.
The discharge is:
- Yellow to yellow-green in color
- Thick — not watery, not thin
- Ropy — can be drawn into strings (this is the most specific qualifier)
- Tenacious — clings to surfaces, difficult to clear
Constitutional Type and Indications
The Hydrastis patient in homeopathic typology is:
- Weak and depleted — often after prolonged illness, surgery, or the "broken-down constitution" of the elderly
- Catarrhal — prone to chronic mucous membrane congestion everywhere
- Hepatic — sluggish liver, poor digestion, bitter taste, yellow-tinged skin
- Cancer diathesis — Hale and later prescribers noted an association with pre-cancerous states and debility from malignancy (used as a supportive remedy, not a treatment for cancer itself)
Primary indications:
| System | Homeopathic Indication |
|---|---|
| Sinuses | Chronic sinusitis with thick yellow post-nasal drip; "dripping down the back of the throat" |
| Nose | Chronic catarrh; thick yellow discharge; nasal obstruction |
| Ears | Thick, tenacious discharge from ears; deafness from eustachian catarrh |
| Digestion | Weak digestion; sinking feeling at epigastrium; bitter taste; constipation with no urge |
| Liver | Jaundice; hepatitis; sluggish liver with foul taste and yellow coating on tongue |
| Female | Leukorrhea — thick, yellow, ropy, tenacious; cervical erosion; uterine prolapse |
| Skin | Ulcers with thick yellow discharge; eczema in folds; old age skin changes |
Modalities
Worse: Morning, open air, cold air, undressing, washing, after eating, during pregnancy
Better: Warmth, warm wrapping, rest, pressure
The Remarkable Parallel
What makes Hydrastis unusual in homeopathy is how closely the homeopathic indications mirror the material-dose (herbal/Eclectic) uses. Most homeopathic remedies are prescribed for symptoms quite different from the plant's known pharmacological effects. But Hydrastis breaks this pattern:
| Use | Eclectic/Herbal | Homeopathic |
|---|---|---|
| Mucous membranes | Primary indication | Primary indication |
| Digestive weakness | Primary indication | Primary indication |
| Liver support | Major use | Major use |
| Eye wash | Traditional use | Ophthalmic catarrh |
| Skin healing | Topical application | Ulcers, eczema |
This convergence may reflect the fact that Hydrastis is most commonly prescribed in low potencies (3X to 6C) where measurable quantities of berberine and hydrastine may still be present, blurring the line between herbal and homeopathic action.
Available Preparations
- Boiron Hydrastis Canadensis 30C — standard single-remedy pellets
- Boiron Hydrastis 6C — lower potency for acute sinusitis
- Mother tincture — available from specialty homeopathic pharmacies (contains active alkaloids; use under professional guidance only)
- SinusCalm and similar combination products — Boiron's multi-remedy sinus formulations frequently include Hydrastis
Part VIII: The Conservation Crisis
From Abundance to CITES
The story of goldenseal's decline is the story of American capitalism meeting a slow-growing forest plant. And the forest lost.
In the early 1800s, goldenseal was described as "abundantly common" throughout the deciduous forests of eastern North America. Dense colonies carpeted the forest floor in the Ohio River valley, the Cumberland Plateau, and the Blue Ridge Mountains. It was so available that settlers used it casually — as a dye, a digestive tonic, and a home remedy for dozens of ailments.
Then the Eclectic movement created industrial-scale demand.
The Timeline of Decline
1850-1880: Eclectic pharmaceutical companies in Cincinnati consume ever-increasing quantities of wild-harvested root. Goldenseal becomes the most commercially valuable botanical drug in America.
1880-1905: Peak harvest era. Professional root diggers strip Appalachian forests systematically. John Uri Lloyd himself warned in 1884 that the plant was becoming scarce in areas where it was once abundant.
1905-1950: Wild populations collapse in much of the original range. Prices rise sharply. The plant becomes rare enough that Eclectic physicians begin recommending substitutes (Oregon grape, barberry).
1950-1990: Herbal medicine revival creates new demand. Wild harvesting continues despite dwindling populations. As late as 1998, an estimated 97.6% of commercially traded goldenseal was wild-harvested — virtually none came from cultivation.
1997: Goldenseal is listed on CITES Appendix II — the Convention on International Trade in Endangered Species. This doesn't ban trade but requires export permits and documentation that harvest is sustainable. The United States, as both the primary range state and the primary consumer, bore particular responsibility.
2000s-present: The IUCN classifies Hydrastis canadensis as Vulnerable — one step below Endangered. Wild populations remain depleted across much of the historical range.
The Math of Overharvesting
Goldenseal's biology makes it uniquely vulnerable to commercial harvesting:
- Slow growth: A colony takes 5-7 years to establish from seed, and 3-5 years from rhizome division to reach harvestable size
- Destructive harvest: The rhizome is the plant. Harvesting kills it. Unlike plants harvested for leaves or bark, there is no sustainable partial harvest
- Habitat specificity: Goldenseal cannot be moved to open fields. It requires intact forest canopy, specific soil conditions, and mycorrhizal associations
- Low seed viability: Seeds require two cold stratification periods and often take 18-24 months to germinate, with germination rates below 50%
When demand exceeds the rate at which populations can replace themselves — and for goldenseal, even modest harvesting can exceed this rate — extinction becomes a mathematical inevitability.
Sustainable Alternatives
Woods-Grown and Wild-Simulated Cultivation: The most promising approach plants goldenseal in its natural forest habitat, providing shade, soil conditions, and companion species that mimic wild conditions. Programs in Vermont, North Carolina, and West Virginia have demonstrated viable commercial production, though yields are lower and timelines longer than conventional agriculture.
United Plant Savers: The conservation organization maintains an "At-Risk" list (goldenseal is a founding member) and operates a 370-acre botanical sanctuary in Ohio where at-risk medicinal plants are cultivated and studied.
Mountain Rose Herbs Conservation: Mountain Rose Herbs has been a leader in sourcing sustainably cultivated goldenseal and funding habitat restoration projects. Their goldenseal products carry organic certification and sourcing documentation.
Oregon Grape as a Substitute: For applications where berberine is the primary desired compound, Oregon grape root (Mahonia aquifolium) offers a sustainable alternative. Oregon grape contains berberine and its own complement of efflux pump inhibitors, grows abundantly in the Pacific Northwest, is not threatened, and can be cultivated easily. For consumers primarily interested in berberine's metabolic effects (blood sugar, cholesterol), Oregon grape or concentrated berberine supplements may be more ecologically responsible choices than goldenseal.
Part IX: Goldenseal Today — Product Recommendations
Choosing Wisely
When purchasing goldenseal products, two considerations should guide your choice: sustainability (is this product contributing to conservation or destruction?) and whole-plant integrity (does it preserve the synergistic chemistry that makes goldenseal more than just berberine?).
Recommended Products
Bulk Herb — Mountain Rose Herbs Mountain Rose Herbs Organic Goldenseal Root Certified organic, sustainably cultivated goldenseal root. Mountain Rose Herbs is a leader in ethical botanical sourcing and has invested directly in goldenseal conservation programs. Available as cut/sifted root for teas and decoctions, or as powder for capsule-filling. This is the gold standard for whole-plant goldenseal.
Berberine Supplement — Dr. Mercola Dr. Mercola Berberine and MicroPQQ Advanced For those primarily seeking berberine's metabolic benefits (blood sugar, cholesterol), a concentrated berberine supplement spares wild goldenseal populations while delivering higher doses. Dr. Mercola's formulation combines berberine with PQQ for mitochondrial support.
Liquid Formulation — MaryRuth Organics MaryRuth Organics Herbal Formulations MaryRuth's line of organic liquid herbal preparations offers high bioavailability and is suitable for those who prefer liquid dosing over capsules. Check their current catalog for goldenseal-containing formulations.
Echinacea + Goldenseal Combination — NOW Foods NOW Foods Echinacea & Goldenseal The classic herbal pairing for immune support, available in standardized capsule form. NOW Foods uses sustainably sourced ingredients and third-party testing for identity and potency.
Echinacea + Goldenseal — Garden of Life Garden of Life mykind Organics Herbal Immune Support USDA Organic, Non-GMO Project Verified echinacea-goldenseal combination. Garden of Life's mykind Organics line uses whole-food extraction methods that preserve the full phytochemical profile.
Homeopathic — Boiron Hydrastis Canadensis 30C Boiron Hydrastis Canadensis Available at most natural pharmacies, Whole Foods, and Amazon. The 30C potency is standard for the homeopathic indications described in Part VII (sinusitis, post-nasal drip, chronic catarrh). Also available in 6C for acute use. Homeopathic preparations use infinitesimal amounts of source material and thus have essentially zero ecological impact.
Sustainable Berberine Alternative — Oregon Grape Root Mountain Rose Herbs Oregon Grape Root For consumers who want berberine-containing herbs without contributing to goldenseal overharvesting, Oregon grape is abundant, easily cultivated, and contains both berberine and its own efflux pump inhibitors. Available as cut root, powder, or tincture.
Safety and Contraindications
Who Should Not Take Goldenseal
Pregnancy: Goldenseal is contraindicated in pregnancy. Berberine stimulates uterine contractions and has shown embryotoxic effects in animal studies. Hydrastine is a known uterine stimulant historically used to control postpartum hemorrhage — its use during pregnancy risks premature labor.
Breastfeeding: Insufficient safety data. Berberine is excreted in breast milk and has caused kernicterus (a form of brain damage from bilirubin displacement) in jaundiced neonates in case reports of berberine-containing herbs. Avoid during lactation.
Children under 2: Not recommended due to the bilirubin displacement risk of berberine.
Drug Interactions
Goldenseal is a potent inhibitor of CYP3A4 and CYP2D6 — two of the most important drug-metabolizing enzymes in the liver. This means it can significantly increase blood levels of drugs metabolized by these enzymes:
| Enzyme | Drugs Affected | Clinical Significance |
|---|---|---|
| CYP3A4 | Statins (simvastatin, atorvastatin), calcium channel blockers, cyclosporine, some HIV protease inhibitors, many benzodiazepines | Potentially serious — can increase drug levels to toxic range |
| CYP2D6 | Codeine, tramadol, metoprolol, many SSRIs (paroxetine, fluoxetine), dextromethorphan | Moderate to significant — altered drug metabolism |
Anticoagulants: Theoretical additive bleeding risk when combined with warfarin or DOACs. Monitor INR if combining.
Antihypertensives: Additive blood pressure lowering effects possible.
Diabetes medications: May potentiate hypoglycemic effects of metformin, sulfonylureas, or insulin. Monitor blood glucose closely if combining.
Duration of Use
Traditional herbalists and the German Commission E recommend goldenseal for short-term use only — typically 2 to 3 weeks continuously, followed by a break of at least 1 week. This is partly based on traditional caution about "cooling" the liver with prolonged use, and partly because berberine may alter gut microbiome composition with chronic administration.
The NTP Carcinogenicity Question
The National Toxicology Program (NTP) conducted a 2-year rodent study (2010) in which goldenseal root powder administered at high doses (25,000 and 50,000 ppm in feed — far exceeding any human dose) was associated with increased liver tumors in rats and mice. The NTP classified goldenseal as providing "clear evidence of carcinogenic activity" in rodents at these doses.
Context matters:
- The doses used were 100-200× higher than typical human supplemental doses, adjusted for body weight
- No epidemiological evidence links goldenseal use to cancer in humans
- Centuries of traditional use have not produced signals of carcinogenicity
- Many substances that are carcinogenic in rodents at extreme doses are safely used by humans (including coffee, vitamin A, and numerous approved medications)
This finding warrants awareness but not alarm. It does, however, reinforce the recommendation against indefinite continuous use at high doses.
Key References
- Barton BS. Collections for an Essay Towards a Materia Medica of the United States. Philadelphia: Edward Earle; 1798.
- Rafinesque CS. Medical Flora; or Manual of the Medical Botany of the United States. 2 vols. 1828-1830.
- King J. The American Eclectic Dispensatory. Cincinnati: Moore, Wilstach, Keys & Co; 1852.
- Lloyd JU, Felter HW. King's American Dispensatory. 18th ed. Cincinnati: Ohio Valley Company; 1898.
- Lloyd JU. Stringtown on the Pike. New York: Dodd, Mead & Company; 1900.
- Hale EM. New Remedies: Their Pathogenetic Effects and Therapeutical Application. 4th ed. 1875.
- Yin J, Xing H, Ye J. Efficacy of berberine in patients with type 2 diabetes mellitus. Metabolism. 2008;57(5):712-717. PMID: 18442638
- Kong W, Wei J, Abidi P, et al. Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins. Nat Med. 2004;10(12):1344-1351. PMID: 15531889
- Dong H, Wang N, Zhao L, Lu F. Berberine in the treatment of type 2 diabetes mellitus: a systemic review and meta-analysis. Evid Based Complement Alternat Med. 2012;2012:591654. PMID: 23512497
- Becker DJ, et al. Red yeast rice for dyslipidemia in statin-intolerant patients: a randomized trial. Ann Intern Med. 2009;150(12):830-839. PMID: 19940377
- Tims MC, Batista C. Effects of multidrug resistance pump inhibitor 5'-methoxyhydnocarpin on the activity of berberine. Antimicrob Agents Chemother. 2011;55(12):5694-5702. PMID: 21157683
- Ettefagh KA, Burns JT, Junio HA, Kaber GW, Cech NB. Goldenseal (Hydrastis canadensis L.) extracts synergistically enhance the antibacterial activity of berberine via efflux pump inhibition. Planta Med. 2011;77(8):835-840. PMID: 21661731
- Zhang Y, et al. Berberine-containing quadruple therapy for initial Helicobacter pylori eradication: an open-label randomized phase IV trial. Medicine. 2017;96(32):e7697. PMID: 28796053
- Chen MJ, et al. Berberine-containing quadruple therapy for Helicobacter pylori infection: a randomized controlled trial. J Formos Med Assoc. 2023;122(10):1053-1060. PMID: 37469024
- National Toxicology Program. Toxicology and Carcinogenesis Studies of Goldenseal Root Powder in F344/N Rats and B6C3F1 Mice. NTP TR 562. 2010.
- Cech NB, et al. Quorum quenching and antimicrobial activity of goldenseal (Hydrastis canadensis) against methicillin-resistant Staphylococcus aureus (MRSA). Planta Med. 2012;78(14):1556-1561. PMID: 22872591
- CITES. Hydrastis canadensis. Appendix II listing. 1997.
- Robbins CS. Comparative analysis of management regimes and medicinal plant trade monitoring mechanisms for American ginseng and goldenseal. Conserv Biol. 2000;14(5):1422-1434.
This article is for research and educational purposes. It does not constitute medical advice. Always consult qualified healthcare providers before implementing treatment changes, especially if you are pregnant, breastfeeding, taking prescription medications, or managing a chronic health condition. Herbal and homeopathic products are not FDA-approved drugs.